Formaldehyde, formalin, paraformaldehyde and glutaraldehyde : What they are and what they do. (from J. Kiernan’s home page)
Copyright (c) 2000 by Today Enterprises.
Article published in Microscopy Today 00-1 pp. 8-12 (2000).
Reproduced with permission.
Formaldehyde, formalin, paraformaldehyde and glutaraldehyde :
What they are and what they do.
John A. Kiernan,
Department of Anatomy & Cell Biology,
The University of Western Ontario,
LONDON, Canada N6A 5C1
Aldehydes are the most commonly used fixatives. They serve to stabilize the fine structural details of cells and tissues prior to examination by light or electron microscopy. Research workers, technicians, pathologists and others who regularly use aldehyde fixatives frequently do not appreciate the nature and properties of these compounds or the reasons for choosing to fix a specimen in formaldehyde, glutaraldehyde or a mixture of the two. Misconceptions are widespread also about formalin and paraformaldehyde, the commercial products from which formaldehyde-containing solutions are made.
Properties of formaldehyde and its polymers
Formaldehyde is a gas. Its small molecules (HCHO, of which the -CHO is the aldehyde group) dissolve rapidly in water, with which they combine chemically to form methylene hydrate, HO-CH2-OH. This is the form in which formaldehyde exists in aqueous solutions ; its chemical reactivity is the same as that of formaldehyde. Methylene hydrate molecules react with one another, combining to form polymers (Fig. 1). The liquid known as formalin contains 37-40% of formaldehyde and 60-63% of water (by weight), with most of the formaldehyde existing as low polymers (n = 2 to 8 in the formula given in Fig. 1). Higher polymers (n up to 100), which are insoluble, are sold as a white powder, paraformaldehyde.
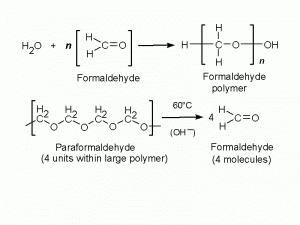
To be useful as a fixative, a solution must contain monomeric formaldehyde (or methylene hydrate, to be pedantic) as its major solute. Dilution with water breaks up the small polymers in formalin. This process is said to take a couple of days if plain water is used, but to be almost instantaneous when formalin is diluted with a buffer solution at physiological pH (Pearse, 1980). Hydrolysis of the polymers is catalyzed by the hydroxide ions present in the slightly alkaline solution (Fig. 1). The big polymer molecules in paraformaldehyde need more energetic treatment. Heating is necessary, as is an added source of hydroxide ions. In one of the earliest paraformaldehyde-derived fixatives (Richardson, 1960) this was sodium sulfite, but the regular practice for at least 35 years has been simply to heat the paraformaldehyde to 60C in water containing the salts used to buffer the solution to pH 7.2 to 7.6.
Formalin contains about 10% methanol, added by the manufacturer because it slows down the polymerization that leads eventually to precipitation of paraformaldehyde. A 4% formaldehyde solution made from formalin therefore contains about 1% methanol. It also contains a small amount of formate ions. These are derived from the Cannizzaro reaction, in which two formaldehyde molecules react together, one being reduced to methanol and the other oxidized to formic acid. Because of this slow reaction, the concentrations of methanol and formate in any formaldehyde solution increase slowly with prolonged storage (Walker, 1964). A solution of formaldehyde prepared from paraformaldehyde, which does not initially contain any methanol, is commonly used in fixatives for electron microscopy and in research applications. Satisfactory ultrastructural preservation is, however, also seen in tissues fixed in buffered formaldehyde generated from formalin (Carson, et al., 1973).
Reaction of formaldehyde with proteins
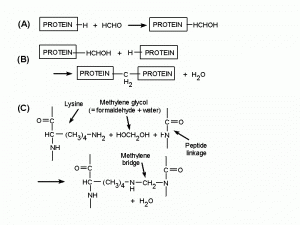
The aldehyde group can combine with nitrogen and some other atoms of proteins, or with two such atoms if they are very close together, forming a cross-link -CH2- called a methylene bridge. Studies of the chemistry of tanning indicate that the most frequent type of cross-link formed by formaldehyde in collagen is between the nitrogen atom at the end of the side-chain of lysine and the nitrogen atom of a peptide linkage (Fig. 2), and the number of such cross-links increases with time (Gustavson, 1956). The tanning of collagen to make leather is comparable to the hardening of a tissue by a fixative (Hopwood, 1969). The fixative action of formaldehyde is probably due entirely to its reactions with proteins. Initial binding of formaldehyde to protein is largely completed in 24 hours (Helander, 1994) but the formation of methylene bridges proceeds much more slowly. Substances such as carbohydrates, lipids and nucleic acids are trapped in a matrix of insolubilized and cross-linked protein molecules but are not chemically changed by formaldehyde unless fixation is prolonged for several weeks.
Practical considerations relating to formaldehyde
This is the most important bit. Formaldehyde penetrates tissues quickly (small molecules), but its reactions with protein, especially cross-linking, occur slowly. Adequate fixation takes days, especially if the specimen must withstand the osmotic and other stresses of dehydration and infiltration with paraffin. Brief fixation in formaldehyde (ideally delivered by perfusion) can stop or greatly reduce autolysis and confer slight hardening and some resistance (but not much) to liquids that are not iso-osmotic with the tissue. This can greatly improve the structural integrity of cryostat and other frozen sections, especially if followed by infiltration with a cryoprotectant such as sucrose (ideally 60% but more usually 15-30%).
When a specimen is dehydrated after only a few hours in formaldehyde, the largely unfixed cytoplasmic proteins are coarsely coagulated. Nuclear chromatin, which contains DNA and strongly basic proteins, is also coagulated by the solvent, forming a pattern of threads, lumps and granules. This is not unlike the appearance induced by fixatives that contain acetic acid, but it is less satisfactory for identifying cell-types on the basis of nuclear morphology. (After adequate formaldehyde fixation, chromatin displays a remarkably even texture, also of little diagnostic value but possibly closer to the structure of the living nucleus.)
Glutaraldehyde solutions
Before 1962 the only satisfactory fixative for electron microscopy was buffered osmium tetroxide. This preserves cellular structure by combining with lipids, especially in membranes, and by insolubilizing some proteins without coagulation, but it is expensive and toxic, penetrates tissues extremely slowly, and extracts much protein and RNA. With the introduction of glutaraldehyde (Sabatini et al., 1962) electron microscopists had a more rapidly penetrating fixative that thoroughly insolubilized proteins and was cheap enough to deliver by vascular perfusion.
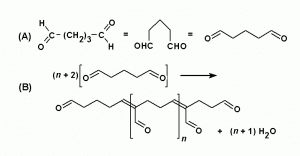
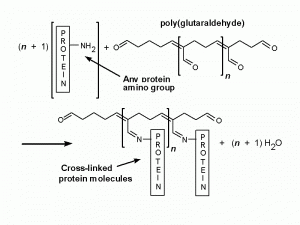
Glutaraldehyde has fairly small molecules, each with two aldehyde groups, separated by a flexible chain of 3 methylene bridges. It is HCO-(CH2)3-CHO. The potential for cross-linking is obviously much greater than with formaldehyde because it can occur through both the -CHO groups and over variable distances. In aqueous solutions, glutaraldehyde is present largely as polymers of variable size (Monsan et al., 1975). There is a free aldehyde group sticking out of the side of each unit of the polymer molecule (Fig. 3), as well as one at each end. All these -CHO groups will combine with any protein nitrogens with which they come into contact, so there is enormous potential for cross-linking, and that is just what happens (Fig. 4). There are also many left-over aldehyde groups (not bound to anything) that cannot be washed out of the tissue.
Practical aspects of glutaraldehyde fixation
Five important points must be remembered when using glutaraldehyde as a fixative for light or electron microscopy.
1. If it’s to be any use as a fixative, especially for electron microscopy, the glutaraldehyde solution must contain the monomer and low polymers (oligomers) with molecules small enough to penetrate the tissue fairly quickly. This means you must buy an “EM grade” glutaraldehyde (25% or 50% solution), not a cheaper “technical” grade. The cheaper stuff, which is for tanning leather, consists largely of polymer molecules too large to fit between the macromolecules of cells and other tissue components.
2. The chemical reaction of glutaraldehyde with protein is fast (minutes to hours), but the larger molecules, especially the oligomers, penetrate tissue slowly. A rat’s brain left overnight in a buffered glutaraldehyde solution and sliced the next day shows a colour change and harder consistency to a depth of 2-3 mm. Objects fixed for a few hours in glutaraldehyde are no longer osmotically responsive (Paljarvi et al., 1979).
3. The free aldehyde groups introduced by glutaraldehyde fixation cause various problems. These include non-specific binding of proteinaceous reagents, notably antibodies, and a direct-positive reaction with Schiff’s reagent). The free aldehydes must be removed or blocked by appropriate histochemical procedures, as described in textbooks (Culling et al., 1985 ; Kiernan, 1999, Ruzin, 1999), before attempting immunohistochemistry, lectin histochemistry, the Feulgen reaction of periodic acid-Schiff staining on glutaraldehyde-fixed material.
4. The thorough cross-linking of a glutaraldehyde-fixed specimen impedes the penetration of fairly large paraffin wax molecules. This makes for difficult cutting and peculiar differential shrinkage artifacts within the specimen. You can stain mitochondria nicely in cells surrounded by obviously abnormal spaces. This is an exaggeration of the inadequacy of formaldehyde and osmium tetroxide as fixatives to precede paraffin (Baker, 1958), and it also highlights the shortcomings of predominantly coagulant fixatives (AFA, Davidson’s, Bouin etc), which preserve the micro-anatomy well but destroy or displace little things like organelles. Fortunately, plastic monomers penetrate glutaraldehyde-fixed tissue adequately. It has been shown that they do not enter every crevice (Horobin & Tomlinson, 1976), but there is enough support to allow the cutting of ultrathin sections for electron microscopy.
5. Immunohistochemistry, which requires as many intact amino acid side-chains as possible, is severely impaired by glutaraldehyde fixation. Nevertheless, clever people have generated antibodies to individual amino acids, that are glutaraldehyde-bound to protein. These allow the detection of soluble amino acid neurotransmitters such as glutamate, GABA and even glycine in presynaptic axon terminals in glutaraldehyde-perfused central nervous tissue (Hodgson et al., 1985 ; Hepler et al., 1988 ; Crooks & Kolb, 1992). Extensive cross-linking also results in the loss or severe reduction of most histochemically demonstrable enzymatic activities, though several are retained after brief fixation (Sabatini et al., 1962).
Mixtures containing formaldehyde and glutaraldehyde
The combination of formaldehyde with glutaraldehyde as a fixative for electron microscopy takes advantage of the rapid penetration of small HCHO molecules, which initiate the structural stabilization of the tissue. Rapid and thorough cross-linking is brought about by the more slowly penetrating glutaraldehyde oligomers. This mixture is associated with the name of Morris J. Karnovsky of Boston. It is an example of a great innovation that was published only in an unrefereed abstract (Karnovsky, 1965). His original mixture contained 4% glutaraldehyde, which was a higher concentration than many people wanted to use (Hayat, 1981). Designations like “half-strength Karnovsky” became common parlance in the 1960s and 1970s. Fixatives of this kind allowed the definitive descriptions of EM-level histology that were accomplished in the 5 or 6 years that followed the introduction of Karnovsky’s fixative, and they are still routinely used.
References
Baker, J.R. (1958). Principles of Biological Microtechnique (Reprinted 1970, with corrections). London : Methuen.
Carson, F.L., Martin, J.H. & Lynn, J.A. (1973). Formalin fixation for electron microscopy : a re-evaluation. American Journal of Clinical Pathology 59 : 365-373.
Crooks, J. & Kolb, H. (1992). Localization of GABA, glycine, glutamate and tyrosine hydroxylase in the human retina. Journal of Comparative Neurology 315 : 287-302.
Culling, C.F.A., Allison, R.T. & Barr, W.T. (1985). Cellular Pathology Technique, 4th ed. London : Butterworths.
Gustavson, K.H. (1956). The Chemistry of Tanning Processes. New York : Academic Press.
Hayat, M.A. (1981). Principles and Techniques of Electron Microscopy. Biological Applications, 2nd ed. Vol. 1. Baltimore : University Park Press.
Helander, K.G. (1994). Kinetic studies of formaldehyde binding in tissue. Biotechnic & Histochemistry 69 : 177-179.
Hepler, J.R., Toomim, C.S., McCarthy, C., Conti, F., Battaglia, G., Rustioni, A. & Petrusz, P. (1988). Characterization of antisera to glutamate and aspartate. Journal of Histochemistry and Cytochemistry 36 : 13-22.
Hodgson, A.J., Penke, B., Erdei, A., Chubb, I.W. & Somogyi, P. (1985). Antisera to -aminobutyric acid. I. Production and characterization using a new model system. Journal of Histochemistry and Cytochemistry 33 : 229-239.
Hopwood, D. (1969). Fixatives and fixation : a review. Histochemical Journal 1 : 323-360.
Horobin, R.W. & Tomlinson, A. (1976). The influence of the embedding medium when staining sections for electron microscopy : the penetration of stains into plastic sections. Journal of Microscopy 108 : 69-78.
Karnovsky, M.J. (1965). A formaldehyde-glutaraldehyde fixative of high osmolality for use in electron microscopy. Journal of Cell Biology 27 : 137A-138A. Kiernan, J.A. (1999). Histological and Histochemical Methods : Theory and Practice, 3rd ed. Oxford : Butterworth-Heinemann.
Monsan, P., Puzo, G. & Marzarguil, H. (1975). Etude du mecanisme d’etablissement des liaisons glutaraldehyde-proteines. Biochimie 57 : 1281-1292.
Paljarvi, L., Garcia, J.H. & Kalimo, H. (1979). The efficiency of aldehyde fixation for electron microscopy : stabilization of rat brain tissue to withstand osmotic stress. Histochemical Journal 11 : 267-276.
Pearse, A.G.E. (1980). Histochemistry, Theoretical and Applied, 4th edn. Vol. 1. Preparative and Optical Technology. Edinburgh : Churchill-Livingstone.
Richardson, K.C. (1960). Studies on the structure of autonomic nerves in the small intestine, correlating the silver-impregnated image in light microscopy with the permanganate-fixed ultrastructure in electronmicroscopy. Journal of Anatomy 94 : 457-472.
Ruzin, S.E. (1999). Plant Microtechnique and Microscopy. New York : Oxford University Press.
Sabatini, D.D., Bensch, K. & Barrnett, R.J. (1963). Cytochemistry and electron microscopy. The preservation of cellular ultrastructure and enzymatic activity by aldehyde fixation. Journal of Cell Biology 17 : 19-58.
Walker, J.F. (1964). Formaldehyde, 3rd ed. New York : Reinhold.
